The lack of protection caused by the total or partial loss of the shell has forced them to develop different defense systems, which they can combine or change, depending on their stage of development. The most effective strategy to avoid falling into the hands of a predator is not to be detected. For this reason, many opisthobranchs use mimicry and crimp or camouflage (by imitating the coloration and form of the substrate, as the first defensive barrier). In our coastline we can find many examples, such as the anaspid Petalifera petalifera, which lives in the leaves of Posidonia oceanica and which is difficult to differentiate due to its greenish coloration. Another example is the sacoglosos, which usually have a color similar to the algae on which they live and feed. In photography it is difficult to differentiate, despite the increase in the image, the two specimens of Elysia viridis on the green background of an algae of the genus Codium. Amongst the nudibranchs, many Doridacea have a similar coloration to the sponge on which they feed.
|
|
|
|
|
|
A typical case of cripsis is that of the dendronotaceo Tritonia nilsodhneri, this small nudibranch lives coiled on the branches of gorgonians of the genus Leptogorgia or Eunicella, from which it copies its color (in the case of gorgonians of red color, the coloration of the nudibranch is almost black, since red is absorbed in the first ten meters of depth) and in addition the ceratas or dorsal-lateral projections imitate the polyps of the gorgonia.
With the same purpose of not being detected, some species, such as the Swiss vaquita Peltodoris atromaculata, present disruptive colors that make their location difficult.
|
|
If they are located, other defensive mechanisms come into play, which can be classified as mechanical or chemical. The first consists of the presence of pointed structures that dissuade the predator when biting its prey, while the second are based on the emission of substances with a bad taste, or that are toxic or with very low pH, sometimes acquired through food

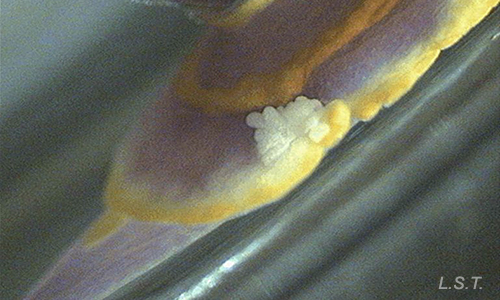
These substances are usually released through glands located at the edge of the mantle (top layer), such as the dermal formations of the mantle of the chromodorids, or in the dorsal part of the mantle.
|
|
|
The species with these defense mechanisms, instead of trying to go unnoticed, usually present very striking colorations (aposematic colorations), which warn their predators of their bad taste, toxicity, etc. However, if all the fish had to learn one by one, that those animals with such striking colors are not good to eat, the few opisthobranchs that survived the lesson would be seriously damaged. Luckily for them, when a fish bites an opisthobranch with chemical defenses, it quickly expels it and the fish in its group, or those around it, quickly learn that these small and colorful animals are not as good as they look.
Taking advantage of these warning signals and the lessons learned from predators, some species associate themselves in what is known as the Müllerian circle (Müllerian mimicry). In the image we can see six species of the Felimare genus of our coastline or littoral, with a similar color pattern that, in the eyes of a neophyte and more like those of a fish, makes them look the same. In addition, one of them, Felimare fountandraui, exploits this circumstance more by lacking chemical defense systems, or mechanisms, and therefore, saving the energy expenditure that this implies (Batesian mimicry).

One type of physical defense, which many Doridacea possess, are the caryophilic tubers, small protuberances surrounded by pointed spicules, which give the animal a scratchy texture that can deter the predator.
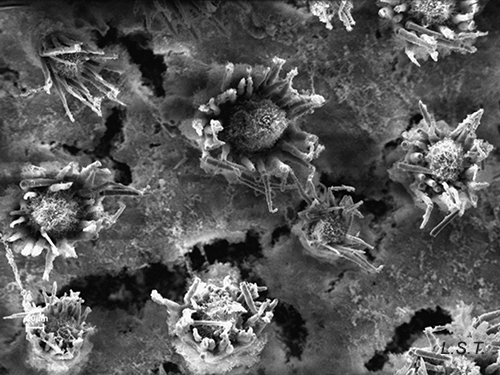
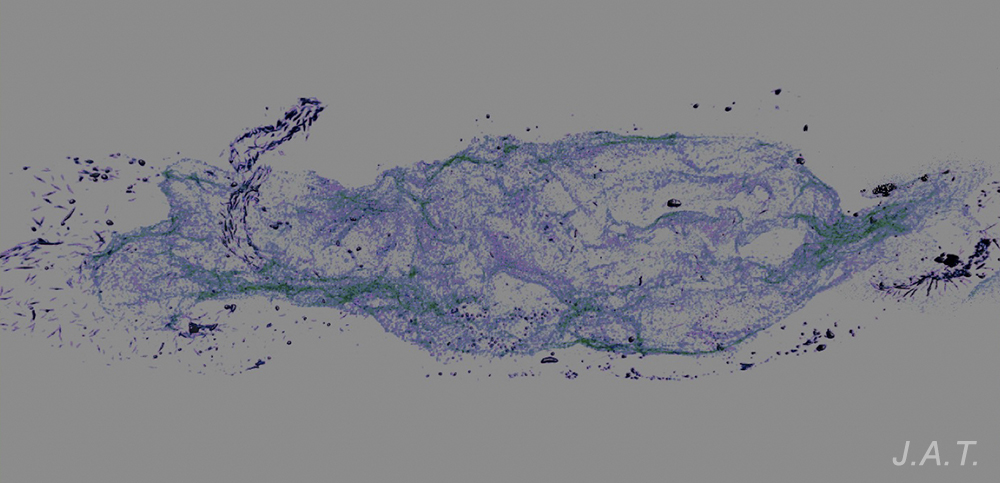
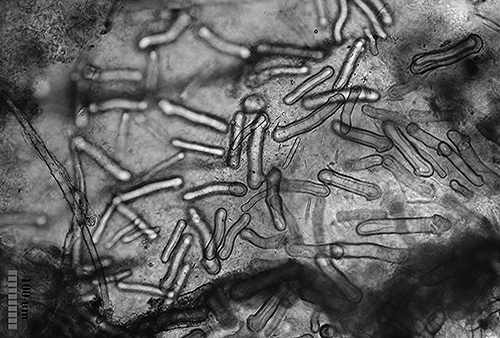
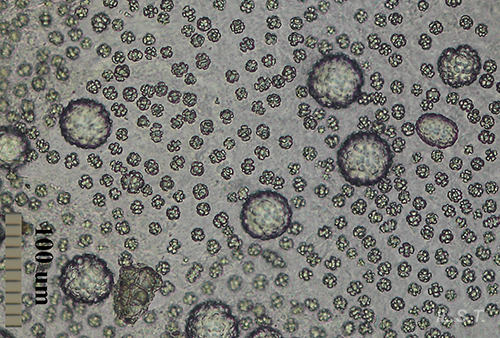
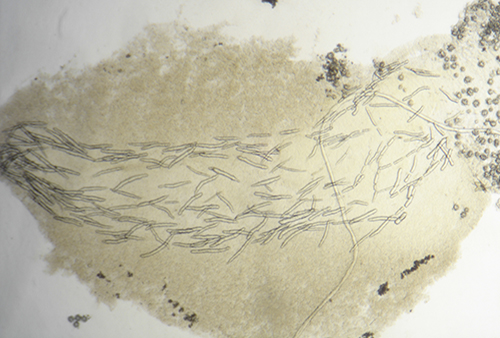
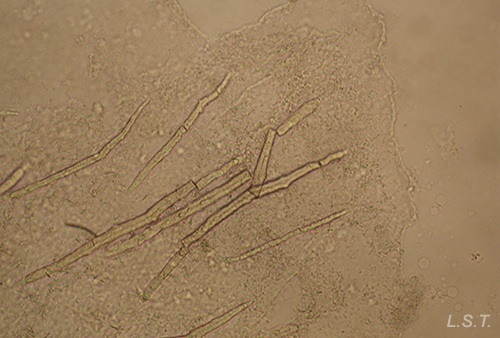
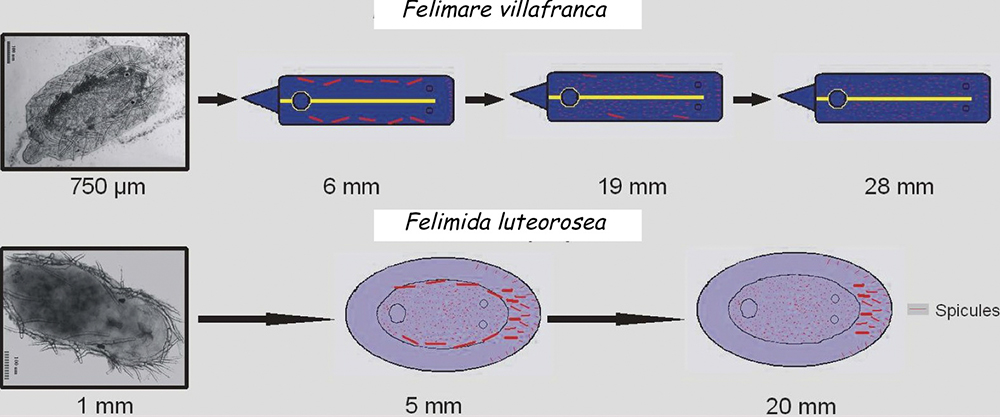
In other cases the spicules, also with a dissuasive function, are embedded in the wall of the body, mainly at the edge of the mantle, rhinophores and the final part of the foot. This type of defense is common in young species of some genus of Doridaceae, in general, aposematics. For example Felimare, Felimida and Dendrodoris. However, in adults of these genera, and with developed chemical systems, the spicules of the mantle have practically disappeared and only those of the rhinophores and the final part of the foot remain. In some species of the genus Felimare: F. bilineata and F. villafranca, the spiky and large-sized spicules of the mantle are replaced by some which are more numerous, less than 100 μm in size. and with blunt ends. Something similar happens in Felimida purpurea, F. luteorosea, F. Krohni and F. binza, but in these species the spicules are spherical, less than 50 μm in size and are embedded in a kind of sheet that is found in the lining of the body. One possible explanation for these changes, in shape and size arrangement throughout their development, is that when they are juveniles and have not yet developed chemical defenses and aposematic coloration, the most effective form of defense, once they are captured, is to deter your predator by pretending to be a food with an unpleasant texture. When they have finally acquired chemical defenses and aposematic or warning coloration, maintaining these dissuasive structures can result in unnecessary energy expenditure.
|
|
|
However, a couple of questions can arise: Why do they preserve the spicules of the rhinophores and those of the end of the foot? and What is the function of the numerous small spicules that they develop at this stage? To answer them we have to look at how a doridáceo behaves before the attack of a predator. Its first reaction is to introduce the gills into the gill cavity, while the rhinophores, which are its means of communication with the environment, remain exposed on the outside. These, like the back of the foot, are very vulnerable to attack, so it does not hurt them to have the spiny feeling provided by the spicules. If the predator continues to insist, the nudibranch is contracted, until it is reduced to half its size at rest, and it is at this moment that the numerous small spicules come into play, since when they come together they form a strong internal armor, which protects the viscera from possible damage caused by the predator, before it spits it out due to the unpleasant chemical defenses.
|
|
|
|
|
|
|
|
Another species in which we have observed an evolution in the spicules throughout its development is the Polycera quadrilineata, a small doridaceous with a striking white and yellow coloration.
The video shows the spicules of a juvenile P. quadrilineata.
In the following image, the differences between a 3 mm sample, a 12 mm one and a 17mm one can be clearly seen.

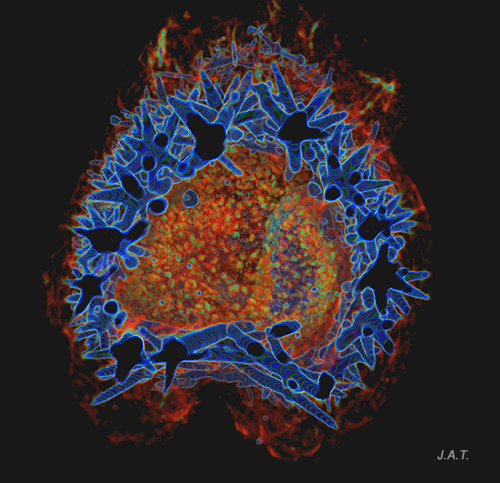
The spicules of Polycera quadrilineata are arranged forming a kind of armor that surrounds the visceral mass, with the pointed parts facing outwards.
Some species, such as Tambja ceutae, escape from their predators, in this case Tyrannodoris europaea, swimming thanks to sudden movements with the body.
A very elaborate system of defense is that presented by aeolides. In general they feed on cnidarians, and not do they outsmart their defense system, but they do it by storing the cnidocytes at the ends of their ceratas or dorsolateral projections, which are connected to the digestive system. Before the attack of a predator, they tend to contract and present the ceratas as if they were the tentacles of an anemone, and they can even get rid of them and later regenerate them. The image shows the similarity between an aeolidid (Facelina bostoniensis) and anemone (Bunodeopsis strumosa), both on Zostera marina.
|
|
The species that do not feed on cnidarians, such as Janolus cristatus, use as a defense their resemblance to the rest of aeolides (Batesian mimicry).

The ability to autotomize parts of their body does not only belong to aeolids, some species of doridáceos also resort to this method and, in case of danger, can partially or completely fragment the edge of the mantle, which serves as a distraction to the predator while seeking refuge.
A very curious case of autotomization is the one that occurs in the species Elysia cf. marginata
Sayaka Mitoh And Yoichi Yusa 2021. Extreme autotomy and whole-body regeneration in photosynthetic sea slugs. Current Biology. DOI:https://doi.org/10.1016/j.cub.2021.01.014
MÁS INFORMACIÓN
VIDEOS
Journey to the center of nudibranch Polycera quadrilineata. Video Prof. Javier Alba-Tercedor.
BIBLIOGRAFÍA
Granada coastline
-Alba-Tercedor, J. & Sánchez-Tocino, L., 2011. The use of the SkyScan 1172 high-resolution micro-CT to elucidate if the spicules of the “sea slugs” (Mollusca: Nudibranchia, Opisthobranchia) have a structural or a defensive function. SkyScan Micro-CT User Meeting. pp. 113-121.
-Alba-Tercedor, J. & Sánchez-Tocino, L., 2012. High-resolution micro-CT of the Anatomy of the Sea Slug Polycera quadrilineata. Microscopy and Analysis. vol 26 (1) pp. 17-18.
-Sánchez-Tocino, L, Tierno de Figueroa, J.M. & Cervera, J.L. 2013. Ontogenetic changes in the spicule formation and their possible role in chromodorid opisthobranchs (Mollusca, Chromodorididae). Marine Biology Research. pp. 357-373.
General
- Avila, C., 1995. Natural products of opisthobranch molluscs: a biological review. Oceanography and Marine Biology Annual Review. 33: 487–559.
- Cheney, K.L.; Cortesi, F.; How, M.J.; Wilson, N.G.; Blomberg,S.P.; Winters, A.E.; Umanzör, S; & Marshall, N.J.Conspicuous visual signals do not coevolve with increased body size in marine sea slugs. Journal of evolutionary biology.
- Cimino, G., Fontana, A. & Gavagnin, M., 1999. Marine opisthobranch molluscs: chemistry and ecology in sacoglossans and dorids. Current Organic Chemistry 3: 327–372.
- Cimino, G. & Ghiselin, M.T., 2009. Chemical Defense and the Evolution of Opistobranch Gastropods. Proceedings of the California Academy of Sciences 60(10): 175–422.
- Cortesi, F. & Cheney, K.L.,2010. Conspicuousness is correlated with toxicity in marine opisthobranchs. Journal of Evolutionary Biology 23: 1509–1518.
- Faulkner, D.J. & Ghiselin, M.T.,1983. Chemical defense and evolutionary ecology of dorid nudibranchs and some other opisthobranch gastropods. Marine Ecology Progress Series 13: 295–301.
- García-Gómez, J.C., Cimino, G. & Medina, A., 1990. Studies on the defensive behavior of Hypselodoris species (Gastropoda: Nudibranchia) Ultrastructure and chemical analysis of mantle dermal formations (MDFs). Marine Biology 106: 245–250.
- García-Gómez, J.C., Medina, A. & Covenas, R., 1991 Study of the anatomy and histology of the mantle dermal formations (MDFs) of Chromodoris and Hypselodoris (Opisthobranchia: Chromodorididae). Malacologia 32(2): 41–46.
- Gosliner, T.M.,2001. Aposematic coloration and mimicry in opisthobranch mollusks: new phylogenetic and experimental data. Bollettino Malacologico. 37: 163–170.
- Gosliner, T.M. & Behrens, D.,1990. Special resemblance, aposematic coloration and mimicry in opisthobranch gastropods. In: Wicksten M, editor. Symposium on the Adaptive Significance of Color in Invertebrates. Sponsored by the American Society of Zoologists. College Station: Texas A. & M. University Press. pp. 127–138.
- Mollo, E., Gavagnin, M., Carbone, M., Guo, Y. & Cimino, G., 2005. Chemical studies on Indopacific Ceratosoma nudibranchs illuminate the protective role of their dorsal horn. Chemoecology 15: 31–36.
-Penney, B.K., 2006. Morphology and biological roles of spicule networks in Cadlina luteomarginata (Nudibranchia, Doridina). Invertebrate Biology, 125(3): pp. 222-232
-Ros, J., 1976. Sistemas de defensa en los opistobranquios. Oecologia Aquatica, 2: pp. 41-77.
-Ros, J.,1977. La defensa en los opistobranquios. Investigación y Ciencia, 12: pp. 48-60.
- Rudman, W.B., 1991. Purpose in pattern: the evolution of colour in chromodorid nudibranchs. Journal of Molluscan Studies. 57: 5–21.
- Thompson, T.E. 1990. Defensive adaptations in Opisthobranchs. J. Mar. Biol.Ass. U.K. 39: 123-134.
- Wägele, H. & Klussmann-Kolb, A.2005. Opisthobranchia (Mollusca, Gastropoda) - More than just slimy slugs. Shell reduction and its implications on defence and foraging.
WEBSITES
LUIS SÁNCHEZ TOCINO